Atomic Force Microscopy

Atomic force microscopy (AFM) is a widely employed method and can be used for force measurements, manipulation, and imaging of surfaces, with the latter being the most common application which was also the topic of this lab course. AFM utilizes a sharp tip which scans over the sample surface to acquire images. The aim of this lab course was to familiarize the students with AFM and give them practical experience in performing measurements and data analysis. In the lab course, the students first examined a calibration grating sample to optimize the feedback parameters for AFM measurements. Then the students investigated the surface of a semicrystalline polymer thin film and analysed the semicrystalline structures.
Broadband Dielectric Spectroscopy
Broadband Dielectric Spectroscopy probes the electrical polarization of a sample in a broad spectral range from Millihertz to Megahertz. This polarization is governed by reorientation motions of polar moieties which enables to trace the dynamics of molecules, of side groups of macromolecules as well as of ions. Mapping the thermal activation of these so-called relaxation times can help to identify structure property relations of material performances ranging from charge transport to dynamics to mechanical stability. We provided a hands-on experience of such investigations on a diverse set of samples to compare among the groups.

Differential Scanning Calorimetry

Differential scanning calorimetry (DSC) is a standard technique used to investigate the thermal behaviour of polymers. With DSC, phase transitions of polymers and proteins can be studied and correlated to the morphology. In this lab course, the students performed DSC measurements of a typical polymer sample to determine the crystallization and glass transition parameters. Also, we demonstrated different calorimetric devices including fast scanning calorimetry (see picture) with its typical applications.
IR spectroscopy
Infrared (IR) spectroscopy is a highly versatile tool in molecular physics with multiple options for refinements, e.g. combining IR spectroscopy with Fast Scanning Calorimetry (FSC) or using grazing angle IR-reflection for interface-sensitive measurements. In this lab course, basic hands-on experiments was be provided by measuring the IR spectra of several samples. By analysing the absorption characteristics which can be understood as the “vibrational fingerprint” of a polymer, the structure of the polymeric sample can be identified. The students were invited to bring their own samples.

Molecular Dynamics Simulations
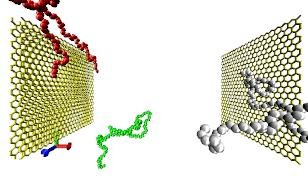
Molecular Dynamics (MD) simulations are a versatile tool to model the properties of polymers, from isolated chains to solutions and melts. In principle they are simply a numerical solution to Newton’s equations of motion, in practice one has to find a model of the polymer system under investigation that may either allow a quantitative prediction of structural or dynamic properties or a qualitative understanding of the behavior of a broad class of polymer systems. The lecture discussed model building as well as some technical background on the numerical approaches. The practical work studied a simple system to gain some feeling on how this approach works.
Monte Carlo Computer Simulations
Monte Carlo computer simulations are an important numerical tool for investigating polymer systems. Instead of solving Newton’s equations of motion as is done in complementary Molecular Dynamics simulations, the Monte Carlo method is based on stochastic “importance sampling” of the state space. This is realized through Markov chains which gives flexibility in the concrete realization of the sampling process and hence often allows for significant optimizations. The aim of this lab course was to provide the students with examples of Monte Carlo codes and give them hands-on experience in using them efficiently. In the lab course, the students first considered a simple toy model and wrote their own computer codes for its simulation and the data analyses. Then they generalized these tools step by step to more involved macromolecular systems.
Nuclear Magnetic Resonance (NMR)

NMR spectroscopy in its simplest form can be conducted on low-field instruments, where one is limited to measure proton spin relaxation times without any spectral resolution. These inform about the overall mobility of the molecular species, and can be used to characterize the composition of soft-matter or polymer systems with significantly inhomogeneous dynamics, such as semicrystalline polymers or food items (e.g. solid starch vs. mobile water in bread). We provided a hands-on experience of such investigations and encouraged the students to bring their own samples.
High resolution NMR on proteins
High resolution NMR spectroscopy allows studies of the structure and dynamics of proteins as well as their interactions with other biomacromolecules. The students got an introduction to high field NMR spectroscopy and will record 1- and 2-dimensional NMR spectra of the protein ubiquitine. During the the hands-on analysis of protein NMR spectra, the students assigned NMR resonances of the protein to the respective nuclei.

Polarized light optical microscopy

Polarized light optical microscopy (POM) is an advantageous method to study crystallization kinetics of polymers. By using crossed polarizers in a light microscope, one can visualize the well-known superstructures formed by synthetic polymers – spherulites, and trace their growth with time. As crystalline lamellae grow radially from the centers of spherulites, the growth of spherulites, thus, contains important information about the growth kinetics of lamellae. The aim of this lab course was to familiarize the students with POM and give them practical experience in performing temperature dependent POM measurements and data analysis. In the lab course, the students performed isothermal crystallization experiments at different temperatures, determine the spherulites growth rate, and analyze the temperature dependence of the growth kinetics.
Single molecule fluorescence spectroscopy
Single molecule fluorescence spectroscopy is a versatile tool that is widely applied in life sciences. We use its unique capabilities to directly relate molecular properties, as size and structure, to spectral properties of conjugated polymers. In the lab course different polymer solutions were investigated to study the effect of solvent quality on the coil size and structured aggregation.

Thermophoretic Single Particles Trapping

Optical manipulation techniques are of great interest in various fields. They allow assembling new materials from the bottom up or provide the means to manipulate and study single molecules. The most common tool for optical manipulations is an optical tweezer, yet this fails to capture single molecules in solution. During this hands-on lab course we introduced a manipulation technique which employs the dynamic generation of heat with the help of optically excited plasmonic nanostructures. The students performed experiments to trap single polymer colloids and single DNA molecules in a thermophoretic trap and studied their dynamics with the help of fluorescence microscopy.
Wide Angle X-Ray Scattering
Wide Angle X-ray Scattering (WAXS) is a typical method to determine the crystalline structures of solids. Simple cubic structures are observed e.g., for polonium, sodium, or calcium, which can be used to explain peak identification and the occurrence of forbidden reflections. In the experimental part, the powder diffractogram of a short alkane (CnH2n+2 with 30<n<60) is measured and compared to a standard polyethylene measurement. The additionally appearing reflections are identified as an indication for the lamellar arrangement of the crystallites. Using Grazing Incidence (GI-) WAXS it can be shown, that the lamellae are arranged parallel to the substrate in thin films.