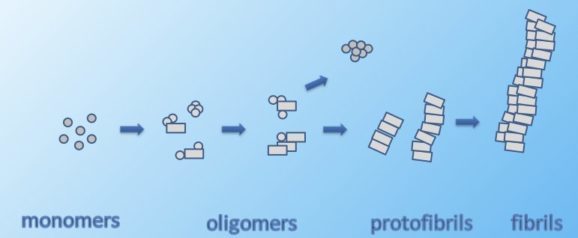
We aim to gain a more detailed understanding of the early prefibrillar growth process of amyloid aggregates and the influence of a constraining environment. In order to reveal the initial pathways of aggregation and disassembly, size distributions of small transient aggregates (named oligomers) are experimentally studied along the time course of fibrillation.
Impact of fluorescence labeling:
Within the complex aggregation process of amyloid peptides into fibrils, early stages of aggregation play a central role and reveal fundamental properties of the underlying mechanism of aggregation. In particular, low molecular weight aggregates of the amyloid β peptide (Aβ) have attracted increasing interest because of their role in cytotoxicity and neuronal apoptosis, typical of aggregation related diseases. To this end, Aβ-peptide chains are functionalized with fluorescent tags, often covalently bound to the disordered N-terminus region of the peptide, with the assumption that functionalization and presence of the fluorophore will not modify the process of self-assembly nor the final fibrillar structure. We could relate the presence of high molecular weight oligomers of Aβ(1-40) to net-attractive, hydrophobic fluorophore-peptide interactions, which are weak in the case of HiLyte 488 and Atto 488. The latter leads for Aβ(1-40) to low molecular weight oligomers only, which is in contrast to Aβ(1-42). The disease relevant peptide Aβ(1-42) displays high molecular weight oligomers even in the absence of significant attractive fluorophore-peptide interactions. Hence, our findings reveal the potentially high impact of the properties of fluorophores on transient aggregates which needs to be included in the interpretation of experimental data of oligomers of fluorescently labeled peptides.
J. Wägele, S. De Sio, B. Voigt, J. Balbach, M. Ott. How fluorescent tags modify oligomer size distributions of the Alzheimer-peptide. Biophys. J. 116, 227-238 (2019)
Funding was provided by the DFG, SFB-Transregio 102, project B12