X-ray scattering of protein solutions
We perform in-house X-ray scattering experiments in the q-range of 0.02-29 nm-1. The wide q-range is realized by varying the sample to detector distance of the 2D-detector. The sample stage contains several capillaries, which can be cooled or heated between -20°C and 180°C.
Example
Scattering intensity of lysozyme at different concentrations:
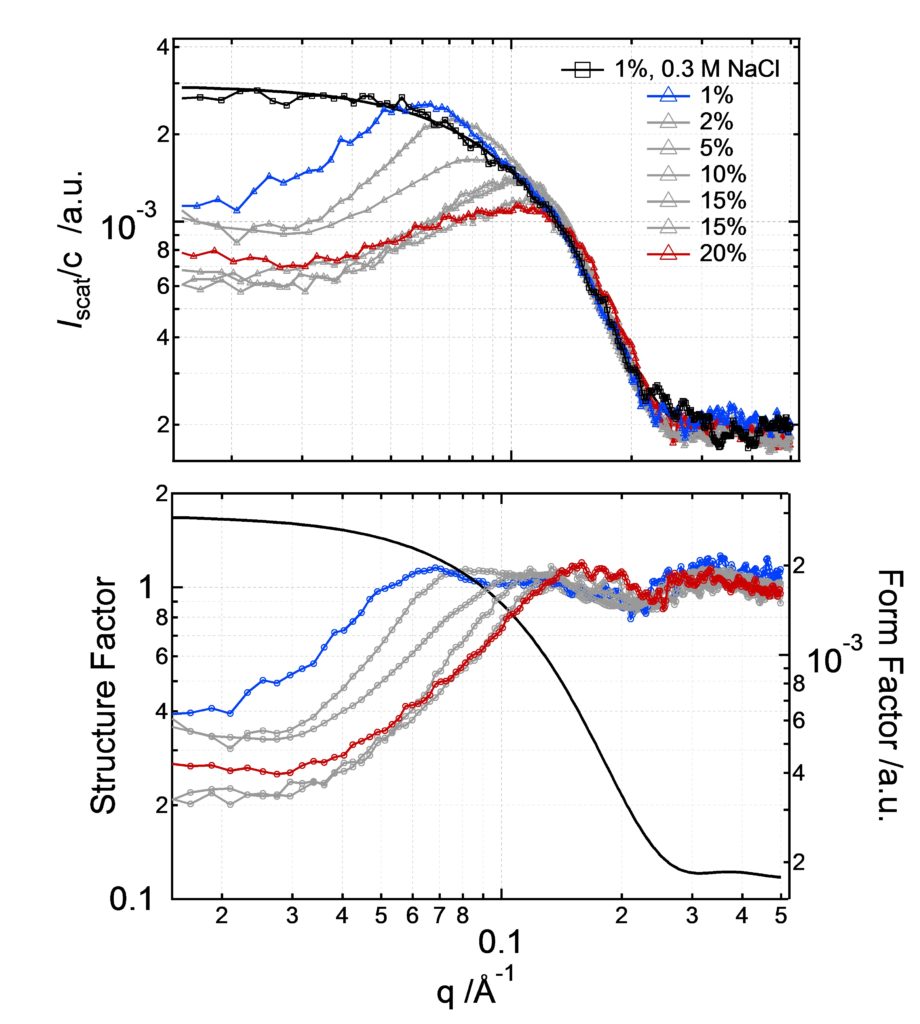
Roos M, Ott M, Hofmann M, Link S, Rössler E, Balbach J, Krushelnitsky A, and K. Saalwächter. Coupling and Decoupling of Rotational and Translational Diffusion of Proteins under Crowding Conditions. J. Am. Chem. Soc. 138,10365–10372 (2016)
X-ray scattering of amyloid fibrils
X-ray diffraction can be used for determining the atomic or molecular structure of crystals or crystal-like samples. Relative distances and absolute positions of the first neighboring atoms can be deduced from the scattering intensity and scattering directions of the X-rays deviated by the atomic electronic densities. X-ray scattering of amyloid fibrils results in a typical os-called cross-beta structure reveal a typical cross-β structure with β-sheets arranged in parallel to the fibril axis and their constituent β-strands perpendicular to the fibril axis. We investigated whether the interaction of fluorophores with the peptide has any impact to the fibrillar structure. We found that the common reflection for the distances of the β-strands (4.75Å) indicates very similar, if not identical, nuclei. With other words, the fluorophores did not affect the basic unit cell of the nuclei, as this structure would propagate during growth. However, the lateral order of the fibrillar bundles was modified, which is depicted by the dye-dependent differences of the other peak at 10 Å.
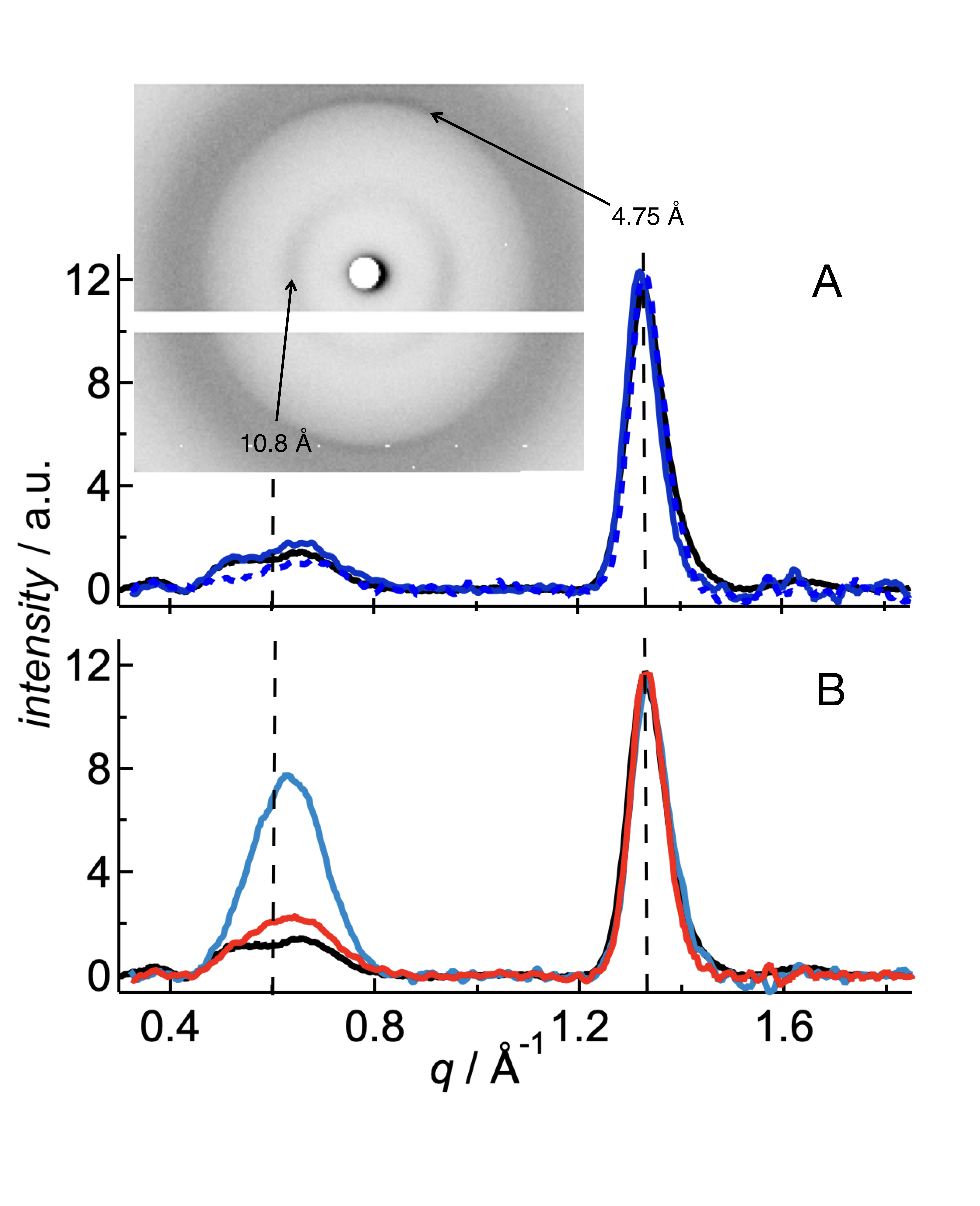 J. Wägele, S. De Sio, B. Voigt, J. Balbach, M. Ott. How fluorescent tags modify oligomer size distributions of the Alzheimer-peptide. Biophys. J. 116, 227-238 (2019)
J. Wägele, S. De Sio, B. Voigt, J. Balbach, M. Ott. How fluorescent tags modify oligomer size distributions of the Alzheimer-peptide. Biophys. J. 116, 227-238 (2019)

